Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Hama, Katsuhiro; Taniguchi, Naoki; Honda, Akira
JNC TN7430 2000-002, 25 Pages, 2001/01
The burial tests of mild steel and pure titanium were performed in a gallery at Tono mine to assess the corrosion resistance of these materials under goundwater environment. Specimens were placed in the container and immersed into groundwater. After the immersion period, the apperance of the surface of these specimens were observed. The corrosion product of mild steel specimen was analysed by various methods. The average corrosion rate of mild steel for 10 years was assessed by the measurement of the weight loss of carbon steel specimen. The results of the test were summerised as follows : (1)The average corrosion rate of mild steel for 10 years was assessed to be 4.36  10
10 mm/y by the weight loss of the specimen. (2)The corrosion product consists of outer porous substance and inner tight corrosion product film. The former contains ferric oxide such as goethite and the latter contains ferrous oxide such as magnetite. (3)The evidence of the initiation of localised corrosion was not observed on the titanium specimens.
mm/y by the weight loss of the specimen. (2)The corrosion product consists of outer porous substance and inner tight corrosion product film. The former contains ferric oxide such as goethite and the latter contains ferrous oxide such as magnetite. (3)The evidence of the initiation of localised corrosion was not observed on the titanium specimens.
Nagano, Tetsushi
Gekkan Chikyu, 22(7), p.429 - 434, 2000/07
no abstracts in English
J. A. BERRY*; M. BROWNSWORD*; D. J. ILETT*; Linklater, C. M.*; Mason, C.*; TWEED, C. J.*
JNC TJ8400 2000-060, 60 Pages, 2000/02
Batch sorption experiments have been carried out to investigate the sorption behaviour of plutonium onto basalt and sandstone from the appropriate rock-equilibrated waters under different redox eonditions. Redox Potentials in solution were controlled by the addition of two reducing agents and one oxidising agent. Thermodynamic chemical modelling was undertaken to interpret the results. The sorption models were based on iron oxide. They adequately reproduced the data for sorption of plutonium onto sandstone, but tended to underpredict sorption onto basalt.
G M N BASTON*; J A BERRY*; M BROWNSWORD*; D J LLETT*; C M LINKLATER*; S W SWANTON*; Tweed, C. J.*
JNC TJ8400 99-078, 72 Pages, 1999/03
A desk study has been carried out to establish the feasibility of measuring the oxidation state of plutonium under near-neutral strongly-reducing conditions. X-ray absorbance spectroscopy appears to be capable of establishing the oxidation state of plutonium sorbed on a suitable substrate. An experimental and modelling investigation has been performed to study the sorption of plutonium onto basalt, mudstone and sandstone under strongly-reducing conditions at three concentrations of carbonate. Appropriate synthetic rock-equilibrated de-ionised water and seawater were used. A model has been developed to describe the sorption of plutonium onto basalt, mudstone and sandstone in de-ionised water and seawater. Predicted R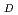 values are generally in good agreement with the observed experimental measurements. The model is based on sorption of plutonium(III) species and assumes iron oxide is the dominant sorbing phase.
values are generally in good agreement with the observed experimental measurements. The model is based on sorption of plutonium(III) species and assumes iron oxide is the dominant sorbing phase.
Sakamoto, Yoshiaki;
Radioact. Waste Manage. Environ. Restorat., 18, p.265 - 280, 1994/00
no abstracts in English
Sakamoto, Yoshiaki; Onuki, Toshihiko;
Radiochimica Acta, 66-67, p.285 - 289, 1994/00
no abstracts in English
Suzuki, Atsuyuki*; Nagasaki, Shinya*
PNC TJ1602 92-001, 75 Pages, 1992/03
For the safety assessment of high level waste disposal system, it is important to understand the migration behaviors of actinides in the near- or far-field. Although the importance of migration of actinides which form the colloids has been pointed out recently, the migration behaviors of colloids are not fully understood. In this study, the migration behaviors of colloids were investigated experimentally by column method. In the experiments, quartz powder was used as the solid. Ferric hydrous oxide colloids were prepared as the real colloids and neptunium(V) formed Np(V)-Fe(III) pseudocolloids by sorption. Some fractions of the colloids eluted with the eluting solution. Also observed were strongly retarded fractions. Non-negligible fractions were found to migrate faster than the eluting solution. These migration characteristics were qualitatively understood in this work.
*; Nagano, Tetsushi; Muraoka, Susumu
Hoshasei Haikibutsu Kenkyu Renrakukai Rombunshu (VI), p.53 - 63, 1991/00
no abstracts in English
Sumiya, Shuichi; Hayashi, Naomi; ; Narita, Osamu
PNC TN8430 91-001, 45 Pages, 1990/12
A radioanalytical method for low level samarium-151(Sm-151) and promethium-147(Pm-147) in environmental samples has been studied for the environmental assessment around nuclear facilities. In this study, we use the separation method with high performance liquid chromatography (HPLC) to determine Sm-151 and Pm-147 in environmental samples such as sea sediments and marine organisms. Samarium-151 and Pm-147 in environmental samples are coprecipitated with other lanthanoids after adding neodymium(Nd). These nuclides are purified by anion exchange methods in methanol-mineral acid media. After the purification, Sm-151 and Pm-147 are separated with HPLC in lactic acid-sodium hydroxide media, and determined with liquid scintillation counting, respectively. The Nd is determined by inductively coupled plasma atomic emission spectrometry (ICP-AES) to correct chemical recoveries of these nuclides. The detection limits for Sm-151 and Pm-147 in this method are about 0.01Bq/sample.
Nagai, Takayuki; Kobayashi, Hidekazu; Hatakeyama, Kiyoshi*; Sato, Seiichi*; Okamoto, Yoshihiro
no journal, ,
no abstracts in English
Nagai, Takayuki; Kobayashi, Hidekazu; Okamoto, Yoshihiro; Inose, Takehiko*; Sato, Seiichi*; Hatakeyama, Kiyoshi*; Seki, Katsumi*
no journal, ,
no abstracts in English